Ⅰ. Overview of Polyvinyl Chloride
Polyvinyl chloride, abbreviated as PVC, is a polymer formed by the polymerization of vinyl chloride monomer (VCM) through a free - radical polymerization mechanism. This reaction is initiated by substances such as peroxides and azo compounds, or under the influence of light and heat. Both homopolymers of vinyl chloride and copolymers of vinyl chloride are collectively referred to as vinyl chloride resins.
PVC is an amorphous white powder with a low degree of branching. Its glass transition temperature ranges from 77 - 90°C, and it starts to decompose at around 170°C. It has poor stability to light and heat. When exposed to temperatures above 100°C or long - term sunlight, it decomposes to produce hydrogen chloride, which further autocatalyzes the decomposition process, causing discoloration and a rapid decline in physical and mechanical properties. In practical applications, stabilizers must be added to enhance its stability to heat and light.
The molecular weight of industrially produced PVC generally ranges from 50,000 to 110,000, with a high degree of polydispersity. The molecular weight increases as the polymerization temperature decreases. PVC has no fixed melting point, it starts to soften at 80 - 85°C, turns into a viscoelastic state at 130°C, and begins to transform into a viscous - flow state at 160 - 180°C. It has good mechanical properties, with a tensile strength of approximately 60 MPa and an impact strength of 5 - 10 kJ/m², as well as excellent dielectric properties.
In its natural color, PVC is slightly yellowish - translucent with a sheen. Its transparency is better than that of polyethylene and polypropylene but worse than that of polystyrene. Depending on the amount of additives used, it can be divided into soft and rigid PVC. Soft products are flexible and tough, with a sticky feel, while hard products have a hardness higher than that of low - density polyethylene but lower than that of polypropylene. Whitening occurs at the bending points.
PVC has the advantages of flame retardancy (with a flame - retardant value of over 40), high chemical resistance (resistant to concentrated hydrochloric acid, 90% sulfuric acid, 60% nitric acid, and 20% sodium hydroxide), good mechanical strength, and electrical insulation.
However, PVC has poor stability to light and heat. Its softening point is 80°C, and it starts to decompose at 130°C. Without heat stabilizers, PVC begins to decompose at 100°C, and the decomposition is faster above 130°C. The decomposition releases hydrogen chloride gas (a toxic gas), causing it to change color from white to light yellow, then to red, brown, and finally black. Ultraviolet light and oxygen in sunlight can cause photo - oxidative decomposition of PVC, reducing its flexibility and eventually making it brittle. This is why some PVC plastics turn yellow and brittle over time.
PVC has stable physical and chemical properties. It is insoluble in water, alcohol, and gasoline, and has low gas and water vapor permeability. At room temperature, it can resist any concentration of hydrochloric acid, sulfuric acid below 90%, nitric acid at 50 - 60%, and caustic soda solution below 20%, showing certain chemical corrosion resistance. It is quite stable to salts but can be dissolved in organic solvents such as ethers, ketones, chlorinated aliphatic hydrocarbons, and aromatic hydrocarbons.
Industrial polyvinyl chloride resin is mainly in an amorphous structure, but it also contains some crystalline regions (about 5%). Therefore, PVC does not have a distinct melting point. It starts to soften at around 80°C, and its heat distortion temperature (under a 1.82 MPa load) is 70 - 71°C. Under pressure, it starts to flow at 150°C and slowly releases hydrogen chloride, causing PVC to change color (from yellow to red, brown, and even black).
The weight - average relative molecular mass of industrial PVC ranges from 48,000 to 480,000, and the corresponding number - average relative molecular mass is 20,000 - 19,500. The weight - average relative molecular mass of most industrial resins is between 100,000 and 200,000, and the number - average relative molecular mass is 45,500 - 64,000. Rigid PVC (without plasticizers) has good mechanical strength, weather resistance, and flame resistance. It can be used alone as a structural material, applied in the chemical industry for manufacturing pipes, plates, and injection - molded products. Reinforcing materials can be added to rigid PVC.
PVC is the plastic with the largest production volume globally among general - purpose plastics and has a wide range of applications. It is widely used in building materials, industrial products, daily necessities, floor coverings, floor tiles, artificial leather, pipes, wires and cables, packaging films, bottles, foamed materials, sealing materials, fibers, etc.
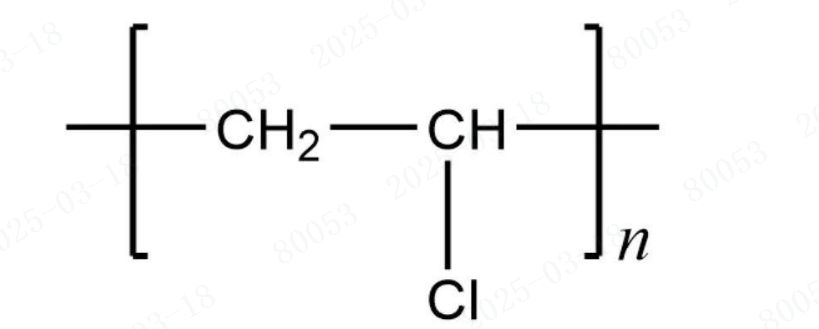
Ⅱ. PVC Classification
By the Size of the Relative Molecular Mass
- General - purpose type: The average degree of polymerization is 500 - 1500.
- High - polymerization - degree type: The average degree of polymerization is greater than 1700.
By Different Morphologies
- Powder form: Used for producing calendered and extruded products.
- Paste form: Used for artificial leather, wallpaper, children's toys, and latex gloves.
By Different Resin Structures
- Compact type: Used for the production of hard products.
- Loose type: Used for the production of soft products.
Ⅲ. PVC Main Raw Materials
Monomer
Vinyl chloride (CH2 = CHCl) is a colorless gas with an ether - like fragrance at normal temperature and pressure and is easy to liquefy.
Sources
- Acetylene - calcium carbide method route.
- Combined method route: Currently the main route for producing vinyl chloride.
- Ethylene oxychlorination method: A new route.
Ⅳ. Production Process of Polyvinyl Chloride
(1) Polymerization Principle and Methods
1. Polymerization Principle
Polymerization mechanism: Free - radical polymerization.
Initiators include oil - soluble azo compounds, organic peroxides, and redox initiation systems.
Chain - growth mode: Head - to - tail connection. Problem: The transfer of the growing chain to the monomer is the main factor affecting the relative molecular mass of the product. This transfer speeds up with increasing temperature.
2. Selection of Polymerization Implementation Methods
Polymerization methods:
- Bulk polymerization: There is a gel effect that causes "auto - acceleration".
- Suspension polymerization: It has the characteristics of simple operation, low production cost, good product quality, good economic benefits, and wide applications, and is suitable for large - scale industrial production.
- Emulsion polymerization: There is a gel effect that causes "auto - acceleration".
- Solution polymerization: It has a high production cost and is rarely used except in the production of special coatings.
Selection principle:
It should be reasonably selected according to product applications, labor intensity, and cost.
(2) Vinyl Chloride Suspension Polymerization Production Process
1. Characteristics and Technological Progress
Suspension polymerization is simple to operate, has a low production cost, good product quality, good economic benefits, and wide applications, making it suitable for large - scale industrial production.
In terms of resin quality, the porosity of PVC resin produced by suspension polymerization has increased by more than 300%. After appropriate treatment, the residual amount of vinyl chloride monomer in the resin has dropped from the original 0.1% to less than 0.0005%. At the same time, improvements in equipment structure, equipment enlargement, and the use of computer - controlled on - line quality control have made the resin quality more stable between batches.
In addition, the development of reactor cleaning technology, large - reactor technology, and residual monomer recovery technology has reduced the number of reactor openings, thereby reducing the release of vinyl chloride monomer. The use of sintering, condensation, or absorption methods for product stripping and waste gas treatment has further reduced the consumption of vinyl chloride monomer.
2. Suspension Polymerization Process Conditions
Vinyl chloride monomer purity: It should be above 99.9%, and the content of other impurities is shown in the following table:
|
Component |
Content |
Component |
Content,% |
Component |
Content,% |
|
Ethylene |
0.0002 |
1-Buten-3-yne |
0.0001 |
HCl |
0 |
|
Propylene |
0.0002 |
Acetaldehyde |
0 |
Iron |
0.00001 |
|
Acetylene |
0.0002 |
Dichloride |
0.0001 |
|
|
|
Butadiene |
0.0002 |
Water |
0.005 |
|
|
Acetylene, when participating in the polymerization, forms unsaturated bonds, deteriorating the thermal stability of the product. The presence of unsaturated polychlorides not only reduces the polymerization rate and the degree of polymerization of the product but also easily generates branches, deteriorating product performance and increasing the number of "fish eyes".
Initiators:
Organic peroxides and azo - type initiators are mostly used in combination, which has a better effect than using them alone. The advantages are uniform reaction speed, more stable operation, good product quality, and enhanced production safety. In industrial production, the polymerization time is generally controlled within 5 - 10 h, and initiators with a half - life t1/2 of 2 - 3 h are selected. If a composite initiator is used, it is best to have one initiator with a t1/2 of 1 - 2 h and another with a t1/2 of 4 - 6 h.
Dispersants:
Gelatin, polyvinyl alcohol, hydroxypropyl methylcellulose, methylcellulose, etc. Gelatin, when used in an amount of 0.05% - 0.2% of the monomer amount, produces resin particles in the shape of ping - pong balls, which are not loose, have non - uniform particle sizes, and have many "fish eyes". Polyvinyl alcohol produces loose, cotton - ball - like porous PVC resin, which has a fast plasticizer absorption rate, good processing and plasticization performance, few "fish eyes", and good thermal stability.
Dispersants (in industry):
Main dispersants: Cellulose - based substances and polyvinyl alcohol (with an alcoholysis degree of 75 - 90%).
Auxiliary dispersants: Non - ionic sorbitols such as monolaurate, monostearate, and tristearate. The combined use of the two also has a good effect.
Water quality and quantity:
Water used is deionized water. The content of chloride ions, iron, and oxygen in the water must be strictly controlled. Excessive chloride ions can cause non - uniform resin particles and an increase in "fish eyes". Iron in the water can reduce the thermal stability of the resin and terminate the reaction. The amount of water used is related to the internal structure of the resin. For the production of compact resin (with gelatin as the dispersant), the mass ratio of monomer to water is 1:1.1 - 1:1.3, for the production of loose resin (with polyvinyl alcohol as the dispersant), the mass ratio of monomer to water is 1:1.4 - 1:2.0. Water plays a role in dispersion, which is beneficial for heat transfer and easy production control.
Oxygen in the system:
Since oxygen has a retarding and inhibiting effect on polymerization, the oxygen in various raw materials and the system should be completely removed.
Other additives:
- pH regulator: The pH value is controlled at 7 - 8. Water - soluble carbonates, phosphates, sodium acetate, and other buffering pH regulators need to be added.
- Anti - sticking agent: Water - bath black, nitroso R salt, and condensates of polyphenols.
- Foam inhibitors (defoamers): Dibutyl phthalate, (un)saturated $C_{12 - 20}$ carboxylic acid glycerides, etc.
- Heat stabilizers, lubricants, etc.
3. Vinyl Chloride Suspension Polymerization Production Process
|
Process Formula (Mass Fraction) |
|
|
Deionized Water |
100 |
|
Vinyl Chloride |
50 - 70 |
|
Suspending Agent (Polyvinyl Alcohol) |
0.05 - 0.5 |
|
Initiator (Diisopropyl Peroxydicarbonate) |
0.02 - 0.3 |
|
Buffer (Disodium Hydrogen Phosphate) |
0 - 0.1 |
|
Antifoaming Agent (Dibutyl Phthalate) |
0 - 0.002 |
Ⅴ. Structure, Properties, and Uses of Polyvinyl Chloride
1. Structure of Polyvinyl Chloride
The molecular chain of PVC contains strongly polar chlorine atoms, resulting in large intermolecular forces. The spatial arrangement of chlorine and hydrogen atoms is basically disordered. The resin also contains a small amount of residual double bonds, branches, and initiator residues.
2. Properties of Polyvinyl Chloride
General properties:
The resin is a white or light - yellow powder, and the hardness of the product can be adjusted by the amount of plasticizer added. Pure PVC has very low water absorption and gas permeability.
Mechanical properties:
PVC has high hardness and mechanical properties, which increase with the increase of the relative molecular mass but decrease with the increase of temperature.
Thermal properties:
The thermal stability of PVC is very poor. It starts to decompose at 140°C and decomposes rapidly at 180°C. It is difficult to burn.
Electrical properties:
The electrical properties of PVC are good, but its electrical insulation is inferior to that of PE and PP. It is generally only used as an insulating material for medium - low voltage and low - frequency applications.
Environmental properties:
PVC can resist most inorganic acids, alkalis, most organic solvents (such as ethanol, gasoline, and mineral oil), and inorganic salts except fuming sulfuric acid and concentrated nitric acid, making it suitable as a chemical anti - corrosion material. However, PVC is prone to degradation, which causes color changes in products, following the order: white → pink → light yellow → brown → reddish - brown → almost black → black.
Processing properties:
① PVC powder resin exists in a granular state. PVC exists in a granular state below 160°C, and the particles break into primary particles above 60°C. The primary particles melt at 190°C.
② The processing stability of PVC is poor. Modification methods include adding heat stabilizers to increase the decomposition temperature above the melting temperature and adding plasticizers to lower the melting temperature below the decomposition temperature. Precise control of the processing temperature is required, and the processing time should be as short as possible.
③ The fluidity of PVC melt is poor, and problems such as melt fracture and rough product surfaces are likely to occur. Processing aids must be added.
④ The friction force between PVC melts and between the melt and processing equipment is large. Internal lubricants with high compatibility or external lubricants with low compatibility need to be added.
⑤ PVC melt is a non - Newtonian fluid. The viscosity can be reduced by increasing the screw speed, but the temperature should be adjusted as little as possible.
⑥ PVC needs to be dried before processing. The conditions are 110°C for 1 - 1.5 h.
⑦ The processing formula of PVC has many components, which need to be fully mixed evenly. Pay attention to the feeding order. Fillers with high oil absorption should be added later to prevent oil absorption, and lubricants should be added later to prevent affecting the dispersion of other components. The mixing temperature is generally 110°C.
⑧ PVC degrades rapidly when it comes into contact with metal ions. Magnetic separation should be carried out before processing, and the equipment should not have rust.
3. Uses of Polyvinyl Chloride
Rigid PVC plastics:
- Pipes: Flexible pipes, rigid pipes, etc.
- Profiles: Doors, windows, furniture, etc.
- Plates: Corrugated plates, solid plates, foamed plates, etc.
- Sheets: Blister products such as packaging boxes.
- Fibers: Window screens, mosquito nets, ropes, etc.
- Bottles: Packaging materials for food, medicine, cosmetics, etc.
- Injection - molded products: Pipe fittings, valves, office supplies, casings, etc.
Soft PVC plastics:
- Films: Greenhouse films, packaging films, decorative films, raincoats.
- Cables: Medium - low - voltage insulation and cable materials.
- Footwear: Rain boots, sandals, shoe soles, upper materials.
- Leather - like materials: Artificial leather, floor leather, wallpaper.
- Others: Soft transparent pipes.
Comparison of PVC with Other Plastic Materials
|
Plastic Material |
Composition |
Characteristics |
Applications |
|
Polyvinyl Chloride (PVC) |
Vinyl chloride monomer |
Corrosion-resistant, highly malleable |
Pipes, flooring, wire sheaths |
|
UPVC |
Unplasticized polyvinyl chloride |
Good corrosion resistance |
Commonly used in manufacturing building applications such as window frames, door frames, and water pipes |
|
Polyethylene (PE) |
Ethylene monomer |
Lightweight, wear-resistant, and low-cost |
Plastic bags, bottles, containers |
|
Polypropylene (PP) |
Propylene monomer |
High-temperature resistance, tough |
Food packaging, containers, automotive parts |
|
CPVC |
Made from polyvinyl chloride (PVC) through a chlorination reaction |
Excellent heat resistance in high-temperature environments |
Commonly used in applications such as high-temperature water pipes and chemical pipelines |
|
Polystyrene (PS) |
Styrene monomer |
Transparent, lightweight |
Foam food containers, toys |
|
Polycarbonate (PC) |
Bisphenol A and propylene oxide |
High strength, high-temperature resistance |
Eyeglasses, water bottles, CDs |
|
Polyphenylene Oxide (PPO) |
Benzene and bisphenol A |
High-temperature resistance, oxidation resistance |
Electronic components, water pipes |
|
Polyethylene Terephthalate (PET) |
Terephthalic acid and ethylene glycol |
Light, transparent, durable |
Beverage bottles, fibers |
|
Nylon (PA) |
Polymer |
Tough, wear-resistant |
Fabrics, brushes, automotive parts |
|
Acrylonitrile Butadiene Styrene (ABS) |
Acrylonitrile, butadiene, styrene |
Tough, impact-resistant |
Toys, electrical appliance casings, televisions |
|
Polytetrafluoroethylene (PTFE) |
Tetrafluoroethylene |
High-temperature resistance, low friction |
Non-stick pans, sealing materials |
Ⅶ. PVC Frequently Asked Questions (FAQs)
Q: What is PVC and how is it produced?
A: Polyvinyl chloride is also known as PVC. It is polymerized from vinyl chloride monomers. Ethylene combines with chlorine to form vinyl chloride, and through polymerization, pure PVC resin is obtained. Depending on the final application, the resin can be processed to obtain different types of PVC compounds, such as rigid and plasticized PVC compounds.
Q: What are the main uses of PVC?
A: Due to its properties and flexibility, PVC is used in many industries.
For example: 1. Pipes, windows and flooring. 2. Cable insulators. 3. Automotive parts and automotive interior parts, binding devices and appliances. 4. Coats, shoes. 5. Goods packaging. 6. Furniture and other household items can be produced as structural or flexible, making it suitable for a variety of products.
Q: What is chlorinated PVC and how is it different from ordinary PVC? Explain.
A: Chlorinated PVC is a modified product obtained through the chlorination process, so it can be called CPVC. In addition, the addition of chlorine affects the heat resistance and stability of the polymer. Since CPVC is designed for different environments, it is usually used in hot water applications and agricultural liquids. However, on the other hand, due to its improved chemical resistance and flame retardant ability, CPVC tends to withstand higher temperature pressure than the latter.
Q: What environmental problems can PVC bring?
A: The environmental problems related to PVC mainly include:
1. Dioxins may be one of the issues of greatest concern to society. The by-products generated during the combustion of PVC and its production process pose a serious threat to the environment. Additives such as phthalates can harm human health and the environment. 3. Due to the presence of other materials, the different components of PVC make it more difficult to recycle. 4. If PVC contains hydrogen chloride, hydrogen chloride may be released during combustion. Fortunately, the PVC industry has found ways to overcome this challenge and make its production more sustainable.
Q: What human health risks may be associated with vinyl chloride?
A: Vinyl chloride, the key substance in PVC, centered around polyvinyl chloride, can bring a variety of health hazards, including:
1. Liver problems, with a high probability of suffering from liver cancer. 2. Nervous or immune system problems. 3. Lung irritation and respiratory problems. 4. Eye and skin irritation upon contact. 5. Exposure to polyvinyl chloride can lead to reproductive and developmental risks, mainly occurring in workers in high-concentration areas or environments, such as homes near PVC manufacturing plants. Finished PVC products around the house do not pose a great risk.
Q: What are the ways of global PVC recycling?
A: The main limiting factor in the age of PVC recycling is the wide variety of additives and mixtures used in different PVC products. Currently, progress has been made in PVC recycling in the following aspects:
1. Mechanical recycling: PVC waste goes through the processes of sorting and grinding, and then is recycled into new products. 2. Chemical recycling: PVC is depolymerized to obtain various chemical components. 3. Energy recycling: Convert unrecyclable polyvinyl chloride into energy. However, these methods do not seem to solve the problem that PVC has a lower rating in recycling than other plastics due to various technical and economic barriers.
Q: In most applications, what potential materials can replace PVC?
A: PVC does have quite a few alternatives, which are currently being researched and used in some industries:
1. Polyethylene, polyethylene (PE) and polypropylene (PP) for pipes and packaging. 2. Ethylene-vinyl acetate (EVA) for flexible applications. 3. Polyurethane (PU) for flooring and interior decoration. 4. Medical materials, such as silicone. 5. Bio-based plastics for part of the packaging and consumer goods. Although it successfully solves some problems caused by the use of PVC, the alternatives also have their own series of environmental and performance problems.





